Clinical practice guideline registration: toward reducing duplication, improving collaboration, and increasing transparency
Registration is a way for researchers to provide key features from their protocol and results which are recorded and published progressively through an online and open platform. In 2007, the International Clinical Trial Registry Platform (ICTRP) based at the World Health Organization (WHO) was established, enabling the full implementation of the clinical trial registry. In 2011, an International Prospective Register of Systematic Reviews (PROSPERO) was developed, so that registration of systematic reviews/Meta-analysis was gradually recognized and applied. Compared with the clinical trials and systematic reviews, clinical practice guidelines (CPGs) are important guidance documents for medical practice. However, prospective registration for CPGs has become relatively late with lacking attention for its importance and significance. The concept, methods and approaches of registration for CPGs have also not been fully discussed by researchers, guideline developers and journal editors for a long time.
With a significant increase in the number of CPGs around the world, duplication of guidelines development on the same clinical topics are common among different countries and inside the same country. Since the outbreak of COVID-19, this phenomenon has become more significant. In order to solve this problem, Annals of Internal Medicine published an article entitled “clinical practice guideline registration: toward reducing duplication, improving collaboration, and increasing transparency” on March 16, 2021. This article was co-authored by the research group of Yaolong Chen combined with experts from Asia, Europe, North America, South America, Australia and Africa. Professor Yaolong Chen is the first and corresponding author of this article.
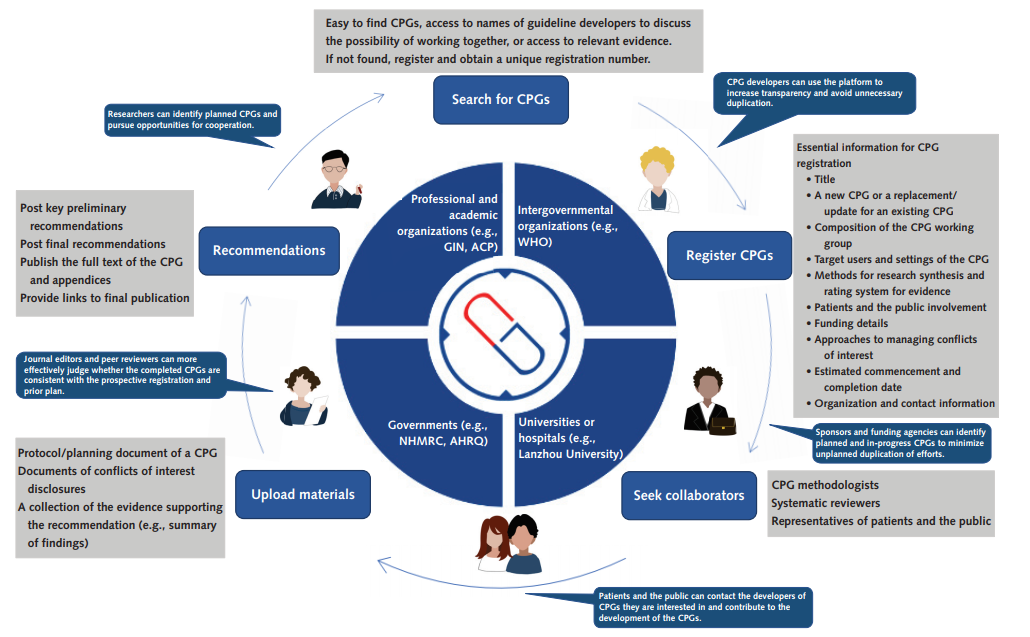
In this article, the authors firstly provided a systematic discussion of the benefits for CPGs registration, including the following five areas: 1) increase transparency and enhance credibility; 2) strengthen cooperation and avoid resource waste; 3) involve patients and the public, incorporate values and preferences; 4) promote dissemination and improve adherence; 5) facilitate peer review and increase feedback. All stakeholders, including CPG developers, sponsors and funding agencies, researchers, patients and the public, CPG users, journal editors and peer reviewers can benefit from guideline registration. Besides this, this article proposed the registration process and operational method of a guideline registry (as shown in the following figure), and put forward the challenges that the guideline registration will face in the future.
At present, there are four platforms for CPG registration in the global. Of these, the German AWMF (Association of the Scientific Medical Societies in Germany) and Australian Government NHMRC (National Health and Medical Research Council) only accept registrations from their own countries. The WHO GRC (Guideline, Review committee) that established in 2007 is an international registry platform, and only responsible for providing regular review of guideline proposals and final version of guidelines to be published by WHO department. Therefore, most of the platforms for CPG registration have some defects in the aspects of global popularization and application. The research group of Yaolong Chen has been devoted to the study of methods and systems of CPG registration, and promoting the dissemination of this concept by cooperating with experts at home and abroad. Then, they established the International Practice Guidelines Registry Platform (IPGRP) in 2014. Compared with the other three registration platforms, although IPGRP was established relatively late, it is open and free for guideline registrants in all countries and regions, which providing a platform for global guideline developers and researchers to search and exchange registration information. In recent years, the number of guidelines that registered on the IPGRP has also been increasing year by year, and up to 450 registrations as of March 19, 2021.
As quoted in this article: three years ago, trials registration was the exception, now it is the rule. We believe that despite the challenges in the future, as the guideline registration system continues to evolve and improve, CPGs will inevitably lead to less duplication, greater collaboration and greater transparency.

